New research suggests booster dose of Novavax NVX-CoV2373 vaccine is effective against SARS-CoV-2 Omicron subvariants

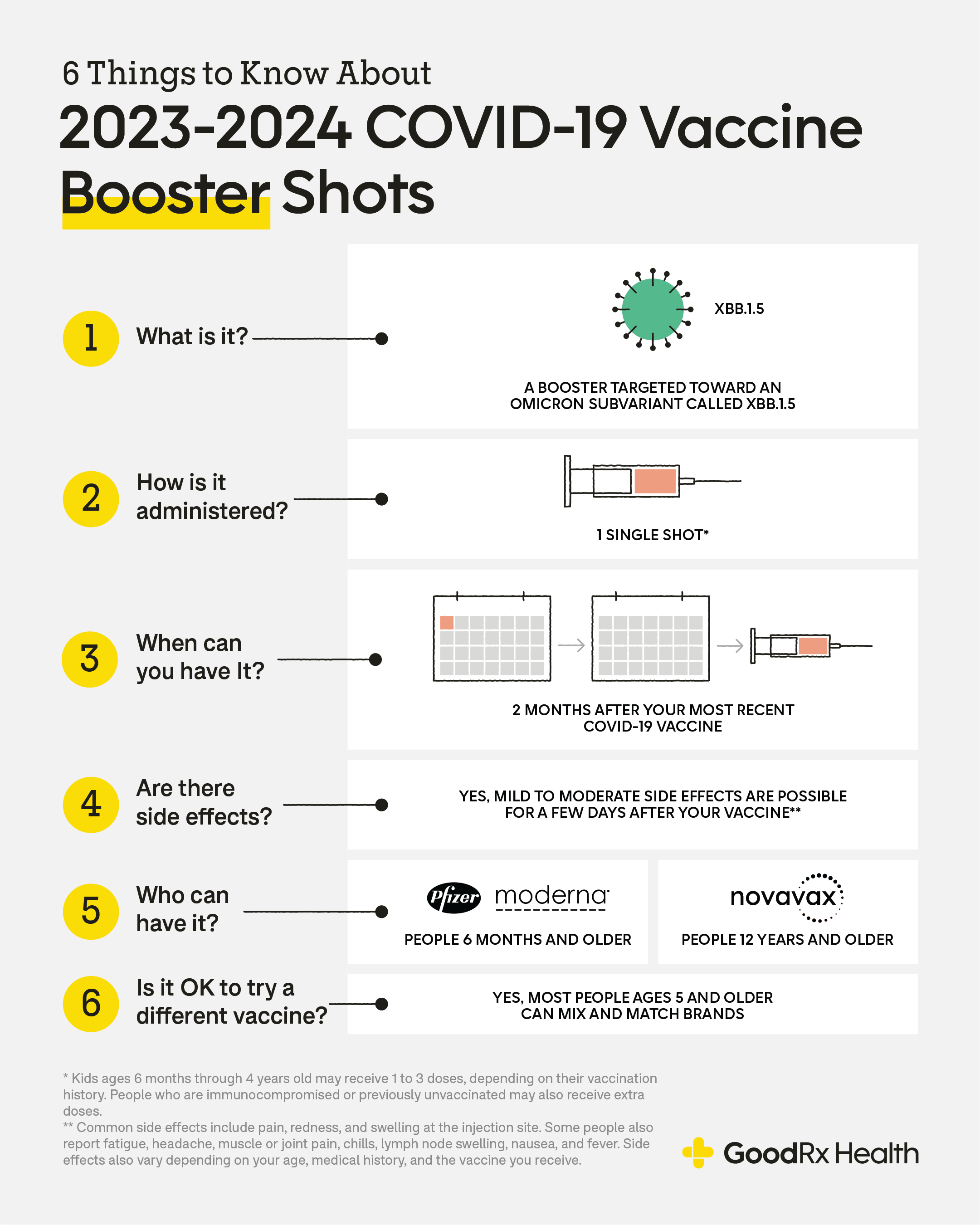
Novavax Prepared to Deliver Protein-based Monovalent XBB COVID Vaccine Consistent with FDA VRBPAC Recommendation for the Fall - Jun 15, 2023
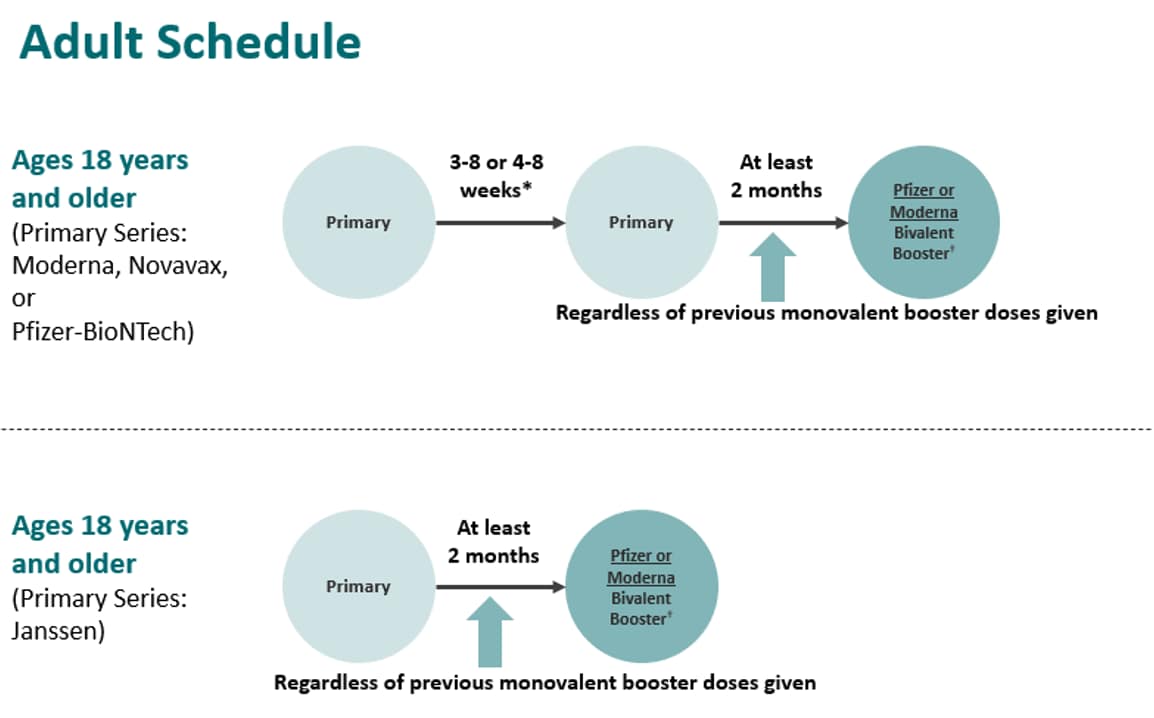
ACIP Evidence to Recommendations (EtR) for Use of Novavax COVID-19 Vaccine Booster Dose for adults ages 18 years and older under an Emergency Use Authorization | CDC
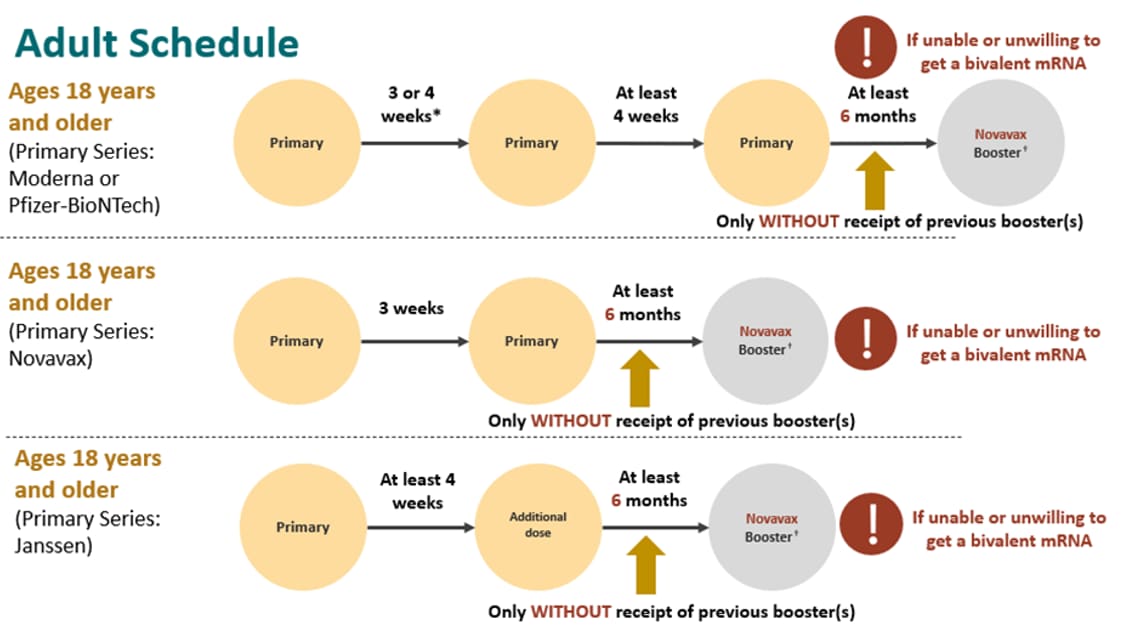
ACIP Evidence to Recommendations (EtR) for Use of Novavax COVID-19 Vaccine Booster Dose for adults ages 18 years and older under an Emergency Use Authorization | CDC

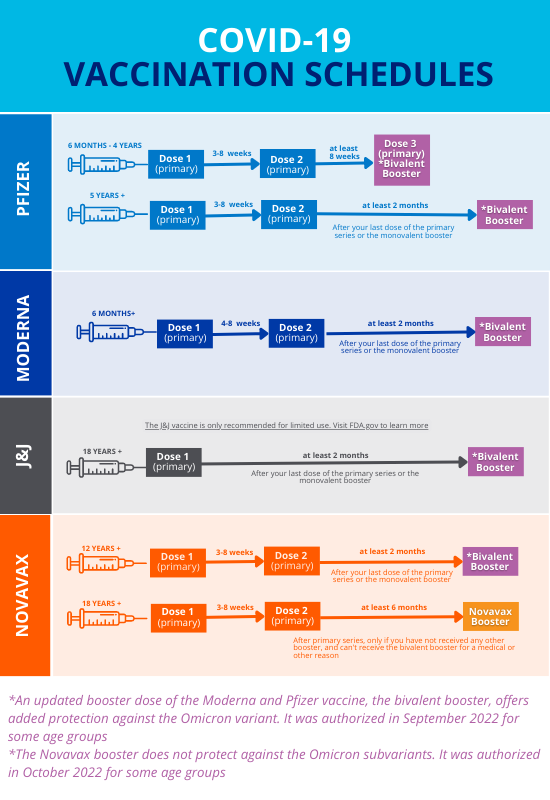



:max_bytes(150000):strip_icc()/Novavax-Files-for-FDA-to-Authorize-Their-COVID-19-Vaccine-Heres-What-to-Know-AdobeStock_476294316_Editorial_Use_Only-2000-3dbc509f2c2a4d5985d8e372cc736433.jpg)