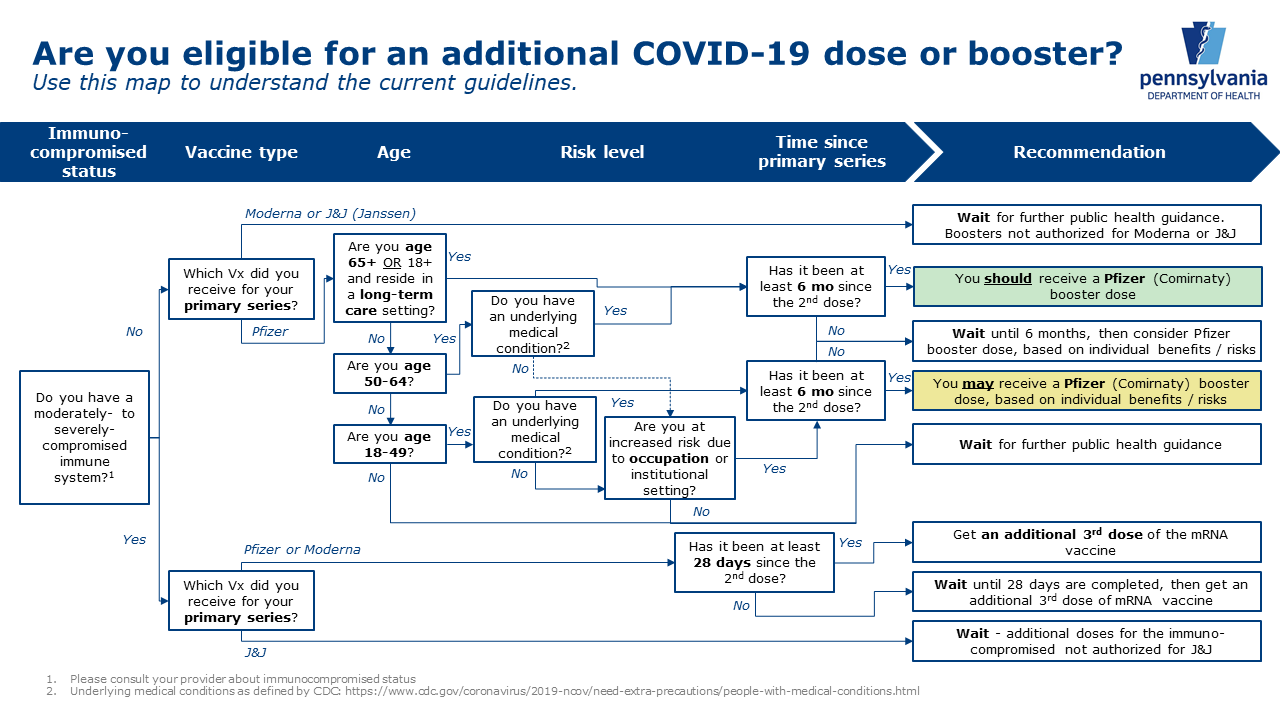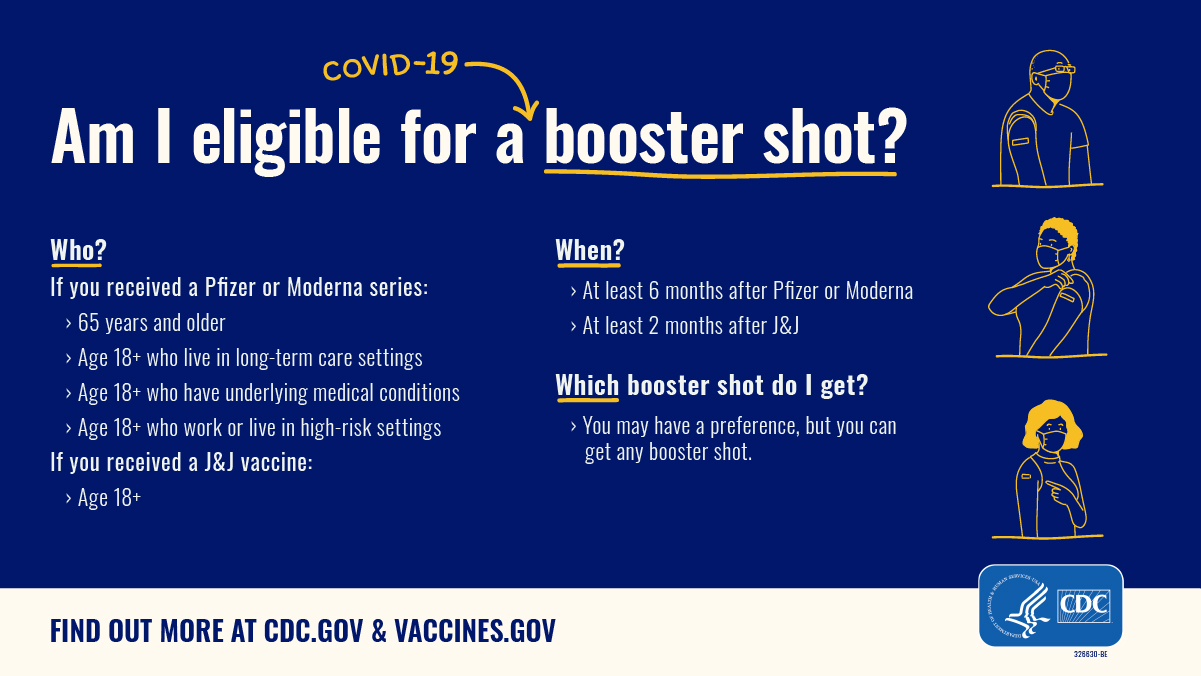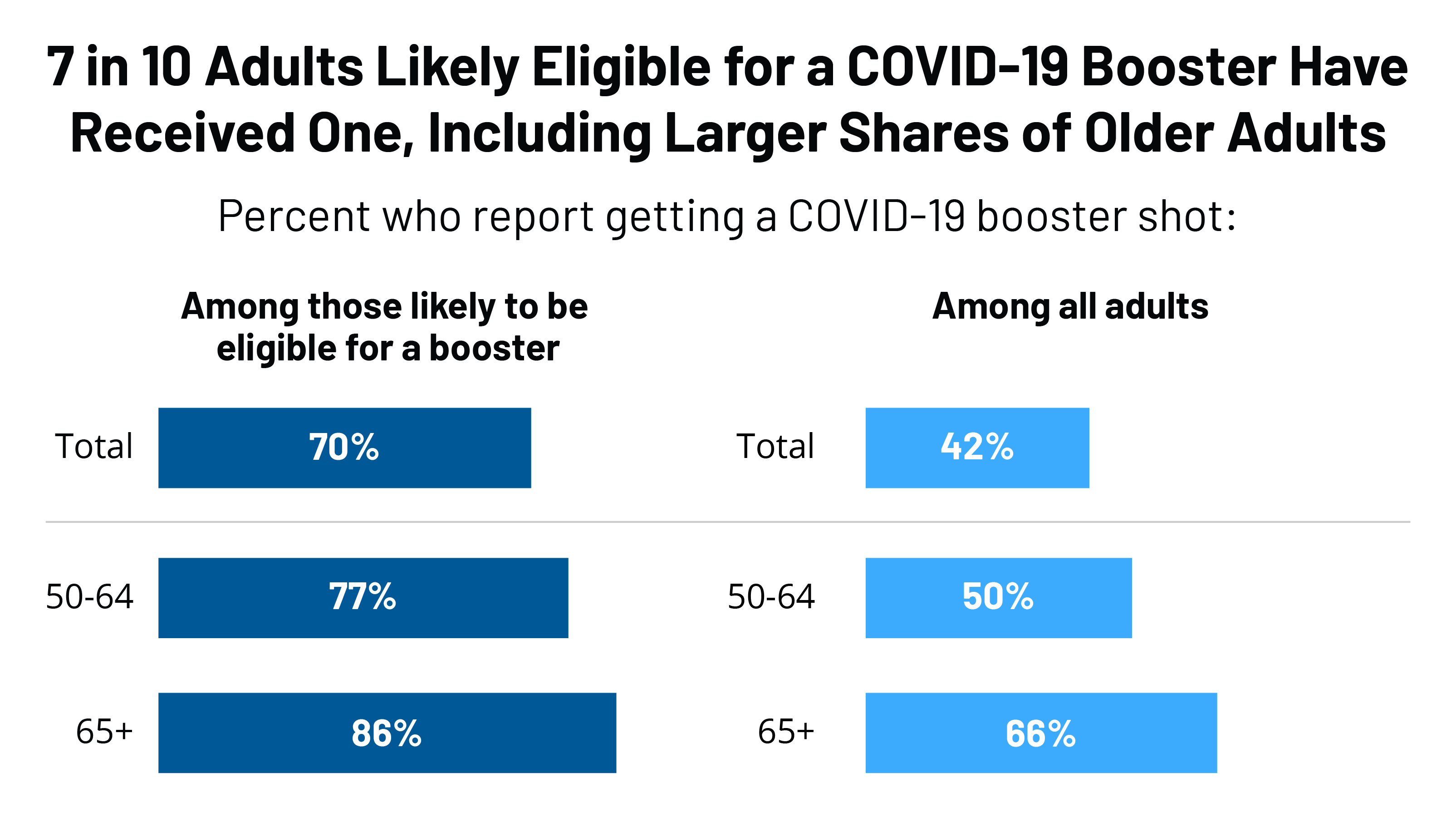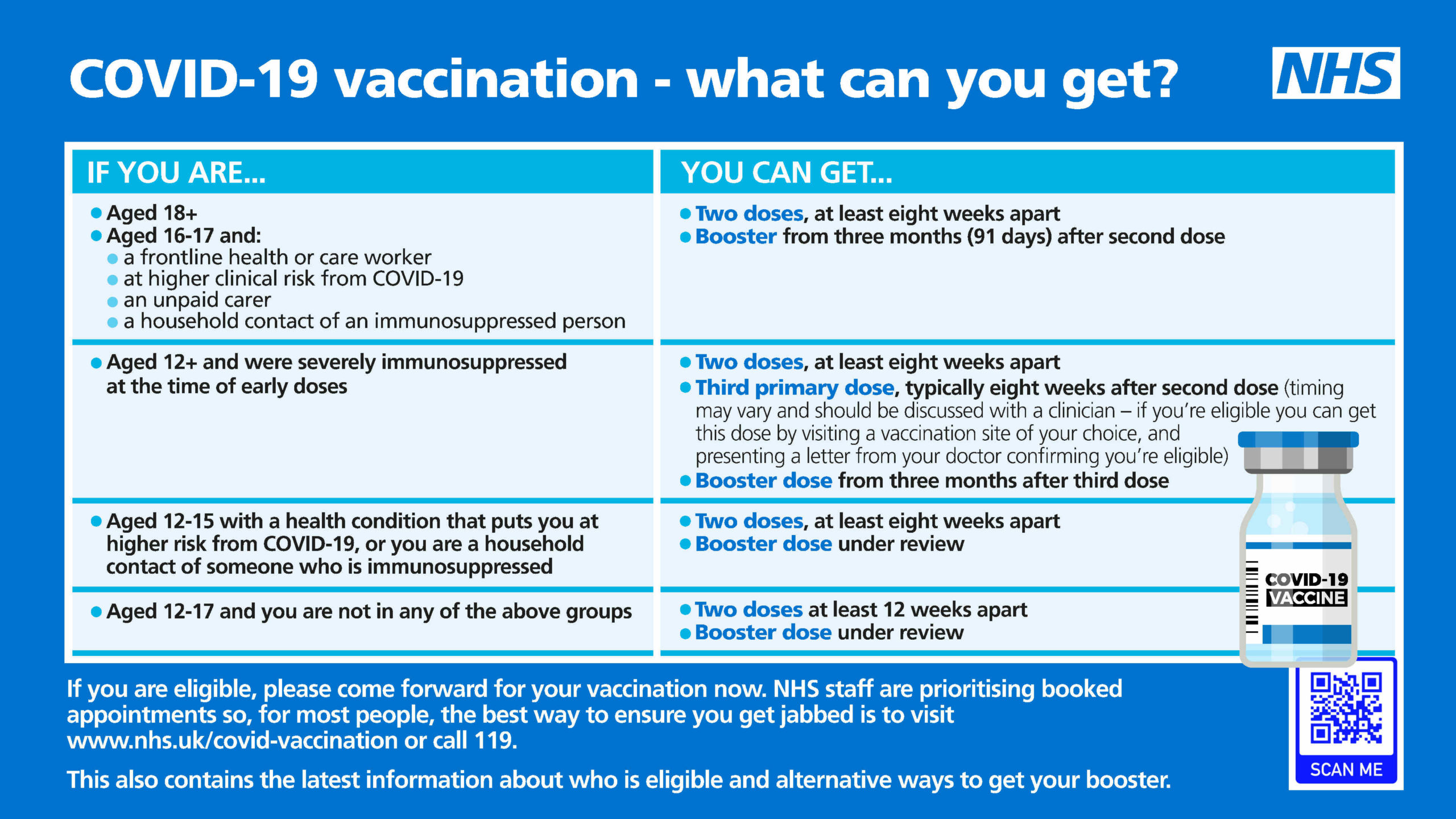
COVID vaccinations: questions and answers about the rollout in North Yorkshire - NHS North Yorkshire CCG

Are you eligible for a COVID-19 spring booster vaccination? Sign up for updates - Shropshire Council Newsroom

Coronavirus (COVID-19) Update: FDA Takes Multiple Actions to Expand Use of Pfizer-BioNTech COVID-19 Vaccine | FDA

Booster shots timeline shortened, Omicron variant detected in Antrim, Charlevoix counties | East Jordan Family Health Center

:max_bytes(150000):strip_icc()/booster-vials-1500x1000-v1copy-446de49e86b2452d9add04b64e9f2790.jpg)